Contents
© The scientific sentence. 2010
|
Bragg's law
1. Bragg's law
The following figure shows x-rays upon the crystal:
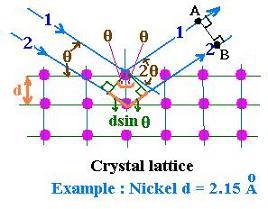
The ray #2 arrives at the point B later. If Δt is the related period of time,
c Δt = 2d sinθ (1.1)
The constructive interferences will occur when
the waves are in phase; that is the 2d sinθ will correspond to the entite
period T = 1/ν ot to an integer number of periods. Then
Δt = n T = n /ν = n λ/c (1.2)
The equation (1.1) and (2.2) give:
nλ = 2d sinθ
That is Bragg's law.
2. Davisson & Germer Experiment
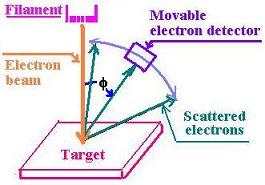 In 1925, C. Davisson and L.H. Germer, working at Bell Telephone laboratories,
investigated the properties of metallic surfaces scattering electrons.
In 1925, C. Davisson and L.H. Germer, working at Bell Telephone laboratories,
investigated the properties of metallic surfaces scattering electrons.
The heated filament send electrons toward the target of Nickel
crystal. For the Nickel, the interatomic distance d is equal to 0.215 nm.
Davisson and Germer found the peak at 500 with Va = 54 V
(the accerating voltage).
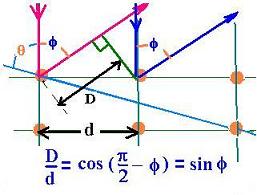
The figure shows the scattering of electrons by lattice plane.
We have:
nλ = D = d sin φ
D is the interatomic distance
The non relativistic kinetic energy of the electron is :
p2/2m = eV
λ = (0.215 nm) sin(500) = 0.165 nm
λ = 0.165 nm
λ = h/p = hc /pc= hc/[2mc2. eVa]1/2
= 1.226 [V/Va]1/2 nm
V0 = 54 V
λ = 0.167 nm
3. Conclusion
Particle exhibited wave properties, consistent with the de Broglie
hypothesis for the wavelengrh of a massive particle.
©: The scientificsentence.net. 2007.
|
|